Managing mycotoxin contaminated feed ingredients is a constant challenge for animal producers. Mycotoxins are produced by both field and storage molds, and – even when fed at low levels – can have a detrimental impact on livestock and poultry performance.1 To make matters worse, grains are frequently contaminated with multiple mycotoxins, and research suggests toxic synergies may exist with certain mycotoxin combinations.2 Absorption of mycotoxins by the gastrointestinal (GI) tract may result in immunological dysfunction, malabsorption of nutrients and ultimately, losses in weight and performance.2 Therefore, when considering mycotoxin management strategies, a key focus area should be on minimizing absorption of toxins by the intestine.
You are viewing United States
- United States
- Canada
- Latin America
- EMENA
- Sub-Saharan Africa
- Russia
- South Asia
- Asia Pacific
- China
- Global
Choose Your Location:
Popular Searches
- Animal Nutrition & Health
- Using Aluminosilicate Anti-Caking Aids to Minimize the Impact of Mycotoxins on Performance
Using Aluminosilicate Anti-Caking Aids to Minimize the Impact of Mycotoxins on Performance
One strategy used for many years to manage mycotoxins is the idea of decontaminating the feed by sequestering the mycotoxin. Sequestration, or neutralization, can reduce the bioavailability of mycotoxins, thereby preventing the toxins from interacting and being absorbed by the intestine.3,4 This practice of neutralization is often completed by incorporating adsorbent feed additives – like hydrated sodium calcium aluminosilicates (HSCAS) – into dietary rations.
Silicates, Aluminosilicates and HSCAS
Characterized by diverse chemical and physical properties, silicates represent approximately 25 percent of all known minerals.5 Silicates are further divided into seven major subclasses based on their structural configuration, two of which are phyllosilicates and tectosilicates. Phyllosilicates – like montmorillonite, bentonite, illite and sepiolite – have a layer cake, or sheet-like, structure; whereas, tectosilicates – like zeolite – are characterized by having an infinite 3D crystal structure framework. Silicates from these two subclasses are often used as anticaking agents in livestock and poultry feed.
Aluminosilicates are silicate minerals where a proportion of the silicon ions are replaced by aluminum. Hydrated sodium calcium aluminosilicates (HSCAS) represent a structurally diverse group of aluminosilicate minerals, many of which have been studied for their ability to sequester mycotoxins.3,4,6 Although generally recognized as efficient binders of aflatoxins, aluminosilicates are typically not considered efficient binders of Fusarium spp. toxins.6 However, it is important for producers to remember that not all aluminosilicates are created equal. Structural differences, absorptive capacity, mineral source and inclusion of additional ingredients can all impact the toxin binding efficiency of a HSCAS mineral.
Evaluating Binding Efficiency of Aluminosilicates
While nothing beats in vivo evaluation, in vitro testing provides quick, reproducible results to evaluate the binding efficiency of different aluminosilicates. However, it’s important to evaluate the binding efficiency under conditions that mimic the digestive process. Specifically, binding efficiency studies should evaluate both the ability of an adsorbent to grab toxins under acidic pH conditions and hold onto the toxins under neutral pH conditions. Measuring only absorption does not provide adequate information to evaluate whether an aluminosilicate will successfully sequester a mycotoxin throughout the GI tract.
Total Binding Efficiency of Enhanced Zeolite
Recently, a two-phase in vitro total binding efficiency (TBE) model was used to evaluate the binding efficiency of an enhanced zeolite mineral flow agent. Enhanced zeolite was dosed at a rate equivalent to 2 lbs. of product per ton of feed. Aflatoxin B1, fumonisin B1, T-2 toxin and zearalenone were tested at a concentration of 3 ppm in all experiments. The TBE of enhanced zeolite for each toxin was determined by comparing percent toxin absorption under acidic buffer conditions (pH 4) with percent toxin desorption under neutral buffer conditions (pH 6.5). Analyses were conducted in triplicate with average adsorption and desorption results used to calculate TBE (Figure 1).7
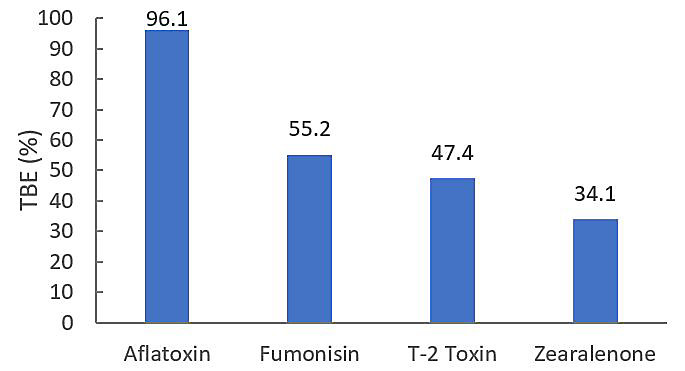
Figure 1. In vitro total binding efficiency (TBE) of enhanced zeolite applied at an equivalent of 2 lbs./ton towards aflatoxin, fumonisin, T-2 toxin and zearalenone. For all toxins, 100% = 3 ppm.
As expected, enhanced zeolite showed extremely efficient binding for aflatoxin. Enhanced zeolite TBE was 55.2% for fumonisin, 47.4% for T-2 toxin and 34.1% of zearalenone. Overall, in vitro TBE of enhanced zeolite was broad-spectrum, showing binding for aflatoxins as well as three of the four Fusarium toxins evaluated in the study. Additionally, binding efficiency of enhanced zeolite was strong even at a low inclusion level.
Conclusions
Use of aluminosilicate feed additives can help producers manage mycotoxin contaminated grains to improve the health and performance of livestock and poultry. However, producers must recognize not all aluminosilicates have equal efficacy. KALLSIL™ – an enhanced zeolite mineral flow agent – is designed to reduce performance losses associated with fungal metabolites found in livestock and poultry operations.8 Importantly, KALLSIL provides broad-spectrum fungal metabolite control to improve feed quality and has minimal binding of vitamins and minerals in feed. More information is available online at Kemin.com/KALLSIL.
References
1Sweeney, M.J. and A.D.W. Dobson. 1998. Mycotoxin production by Aspergillus, Fusarium and Penicillium species. Intern. J. Food Microbiol. 43:141-158.
2Wan, L.Y.M., P.C. Turner, and H. El-Nezami. (2013). Individual and combined cytotoxic effects of Fusarium toxins (deoxynivalenol, nivalenol, zearalenone and fumonisins B1) on swine jejunal epithelial cells. Food and Chemical Toxicology, 57:276-283.
3Ramos, A.J., J. Fink-Gremmels, and E. Hernández. (1996). Prevention of toxic effects of mycotoxins by means of nonnutritive adsorbent compounds. J. Food Protection, 59(6):631-641.
4Vila-Donat, P., S. Marín, V. Sanchis, and A. J. Ramos. (2018). A review of the mycotoxin adsorbing agents, with an emphasis on their multi-binding capacity, for animal feed decontamination. Food and Chemical Toxicology, 114:246-259.
5"Mineral - Silicates." britannica.com. Archived from the original on 25 October 2017. Retrieved 8 May 2018.
6Jouany, J.P. (2007). Methods for preventing, decontaminating and minimizing the toxicity of mycotoxins in feeds. Anim. Feed Sci. and Tech., 137:342-362.
7Comparative in vitro binding efficiency of two common feed additives towards mycotoxins present in livestock and poultry feeds, INF-2018-00069.
8Tilley, J.E.N., et al. (2017). Efficacy of feed additives to reduce the effect of naturally occurring mycotoxins fed to turkey hen poults reared to 6 weeks of age. Poultry Science, 96:4236-4244.
Have a Question?
If you have a question about our products or services, or just want more information, fill out the form below and someone on our team will be in contact with you.
- California Residents
- California Supply Chain Act
- Canada Forced Labor and Child Labor Report
- Email Disclaimer
- GDPR Personal Data Addendum
- General Terms & Conditions for Vendors
- Global Environmental Policy Statement
- Indirect Cost Estimates
- Kemin Terms & Conditions
- Modern Slavery Act Transparency Statement
- Privacy Policy
- Change Cookie Consent
- Animal Welfare Statement
© Kemin Industries, Inc. and its group of companies All rights reserved. ® ™ Trademarks of Kemin Industries, Inc., USA
Certain statements may not be applicable in all geographical regions. Product labeling and associated claims may differ based upon government requirements.
Thank you for visiting Kemin.com.
Your questions and feedback are important to us. Let us know how we can help you learn more about Kemin, our products, our services or our website.
Interested in starting a career with Kemin?
