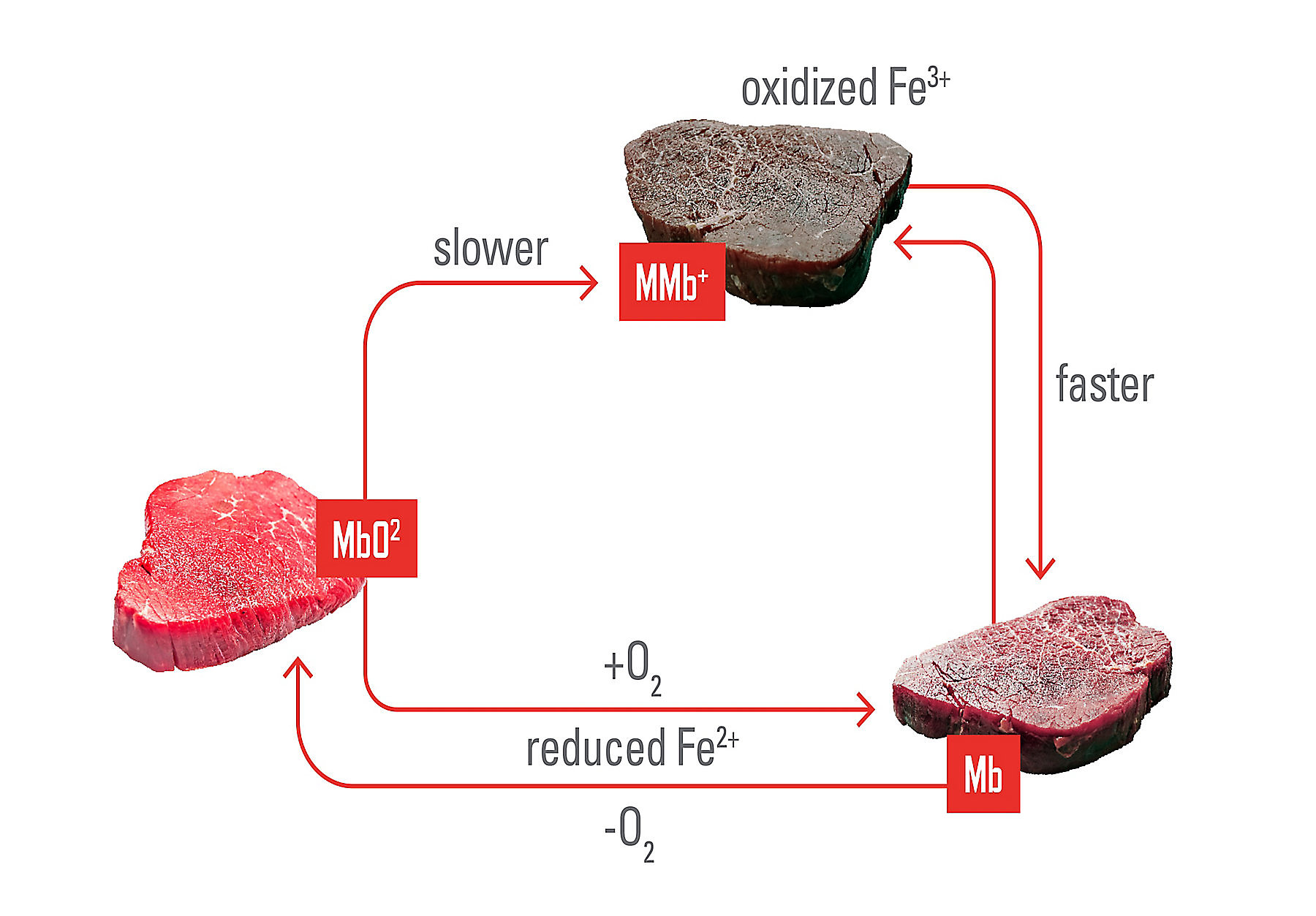
Role of antioxidants in meat colour and oxidative stability
Oxidative reactions are among the main factors influencing both lipid stability and meat colour deterioration during processing and storage. Several naturally derived ingredients commonly studied in meat systems act through complementary antioxidant mechanisms, contributing to colour stability either directly, by interacting with myoglobin, or indirectly, by limiting lipid oxidation and secondary reaction products.⁶
Plant extracts rich in phenolic compounds
Plant‑derived extracts such as rosemary (Rosmarinus officinalis), green tea (Camellia sinensis), and olive (Olea europaea) have been widely investigated for their antioxidant activity in meat applications. Their effectiveness is largely attributed to their phenolic composition, which enables free‑radical scavenging and metal‑chelating activity.⁶
Rosemary extract
Rich in phenolic diterpenes such as carnosic acid and carnosol, rosemary extract is known for its ability to inhibit lipid peroxidation. By slowing lipid oxidation, it can indirectly support colour stability, as lipid‑derived oxidation products contribute to myoglobin oxidation and metmyoglobin formation.⁶
Green tea extract
Green tea contains catechins, including epigallocatechin gallate (EGCG), which exhibit strong antioxidant activity. These compounds act primarily as radical scavengers and have been studied for their capacity to reduce oxidative reactions in raw and processed meat systems.⁶
Olive extract
Olive‑derived extracts contain phenolic compounds such as hydroxytyrosol and related derivatives. These compounds have been associated with antioxidant effects in meat matrices, particularly through their ability to limit oxidative chain reactions and interact with pro‑oxidant metal ions.⁶
Ascorbic acid and acerola cherry derivatives
Ascorbic acid plays a distinct role in meat colour stability due to its reducing properties. It can delay discolouration by helping maintain the heme iron of myoglobin in its reduced (Fe²⁺) state, thereby slowing the conversion to metmyoglobin.⁴
Acerola cherry (Malpighia emarginata) is a natural source of ascorbic acid and has therefore been studied as an alternative way to introduce reducing activity into meat formulations. By supporting redox balance, ascorbic acid and its naturally derived sources may contribute to improved colour stability during storage and retail display.⁴
Complementary antioxidant mechanisms
In addition to radical scavenging and reducing activity, some formulation components contribute to oxidative stability through metal ion chelation. Chelating agents such as citric acid can bind pro‑oxidant metals, thereby reducing their catalytic role in oxidation reactions.⁶
Taken together, these complementary mechanisms highlight how ingredient selection can influence oxidative reactions, lipid stability, and meat colour during processing and storage. A science‑based understanding of antioxidant function supports informed formulation strategies aimed at maintaining visual quality across shelf life.